 Blacklegged ticks are collected in vials and tested for Lyme in November 2014. (Photo courtesy of Graham Hickling)
Blacklegged ticks are collected in vials and tested for Lyme in November 2014. (Photo courtesy of Graham Hickling)
In the summer of 1975, a group of concerned mothers in Connecticut notified public-health officials about several cases of children displaying rheumatoid arthritis-like symptoms. Working with Allen Steere, a young rheumatologist at Yale University School of Medicine, authorities traced the cases to parks and gardens where they could verify the presence of ticks, which they assumed had played a role in transmitting a novel infection. In 1976, they named the condition Lyme disease after the small town where parents had observed the first cluster of infections. Soon after, in 1981, scientist Willy Burgdorfer identified the pathogen responsible for the infection: a spirochetal bacterium later named Borrelia burgdorferi in his honor.
The swift identification of the disease-causing pathogen created the hope throughout the 1980s and ’90s that the development of diagnostic tools and discovery of therapeutic cures were imminent. Fifty years later, however, Lyme disease now infects close to half a million Americans annually, and there is still no easy way to diagnose, let alone cure, patients. In the United States, Lyme accounts for 82 percent of all tick-borne-disease cases and 77 percent of all vector-borne-disease cases (or diseases resulting from infections transmitted by blood-feeding insects and parasites) across all 50 states. Symptoms affect multiple systems in the body, can be easily confused for symptoms of other conditions, and are often debilitating. Depending on the location of the infection, patients may suffer from symptoms including extreme fatigue; cognitive challenges, such as brain fog and recall deficits; extreme mental-health issues, such as panic attacks and suicidal thoughts; neurological problems; severe joint pain; and heart inflammation.
In January 2024, the US Department of Health and Human Services (HHS) published the first-ever national strategy to address the global threat of vector-borne diseases. Its arrival is a milestone because it identifies the needs of the community of patients and is a guide for the allocation of resources that fuel scientific research. Ideally, however, the government’s recognition of the seriousness of the threat of vector-borne diseases for public health would have happened decades earlier.
As someone whose life has been disrupted by Lyme disease, I have experienced society’s slow and disorganized response to Lyme and other complex chronic diseases. At the same time, I’ve had the chance to witness the work of remarkable people committed to making a difference in the lives of patients. Today, I contribute my perspective as an educator in innovative approaches to complex and often intractable societal problems to learn from past community-led advocacy efforts, make sense of the existing gains and remaining gaps, and illuminate the path forward.
In what follows, I examine the reasons why both the scientific community and the government lost their initial momentum to find effective diagnostic and therapeutic solutions to Lyme disease. I chronicle how a group of connected individuals launched an innovation movement and mobilized the government to reboot the United States’ approach to the important threat of vector-borne diseases. Lyme disease is only a small manifestation of the much larger public-health issue of infection-associated complex chronic diseases, so I conclude by highlighting the opportunity to think expansively and join forces across a broad range of long-haul conditions.
A Contested and Underfunded Illness
When the Connecticut cluster of Lyme cases emerged, scientists around the world were advancing evidence-based medicine and broadening the scope of biomedicine by codifying biological disorders and standardizing care. But in the case of tick-borne infections, the scientific effort was no match for the range of pathogens and for the biological complexity of the multistage and multisystemic pathogenesis of these diseases.
Borrelia is an evolutionarily complex bacterium with sophisticated faculties that leverage the biology of successive mammalian hosts. The intricate host-pathogen interactions are difficult to reproduce in vitro and therefore make the bacterium challenging to study. Its spread is facilitated by macro-environmental factors—from climate change to deforestation and other land-management practices—that disrupt natural ecosystems and support the abundance and geographic expansion of host animals.
Scientists were in consensus that the first stage of Lyme infection was bacterial in nature. However, they were split on explanations of the later stages of the disease. In the 2000s, the scientific and medical communities established two distinct models for late-stage Lyme disease: post-treatment Lyme disease syndrome (PTLDS) and chronic Lyme. The two models diverge at the second stage of the disease, which explains the symptoms experienced by some patients after they receive antibiotics to treat the initial infection. The difference resides in opposing hypotheses about whether Borrelia can persist in humans after antibiotics treatment: The PTLDS model says it cannot, while the chronic Lyme model says it can. Based on their experience of the chronic nature of the symptoms, most patient advocates supported the chronic Lyme model.
The conflicting perspectives resulted in two standards of care with different philosophies about the use of antibiotics. In 2000, 2006, and 2020, the Infectious Diseases Society of America (IDSA) published guidelines recommending a short and unique course of doxycycline, an antibiotic used widely to treat bacterial infections. These recommendations leaned on scientific studies that concluded that antibiotics eliminate Borrelia. As a result of these findings, IDSA viewed late-stage Lyme disease as a medically unexplainable illness without measurable biological markers.
Conversely, the International Lyme and Associated Diseases Society (ILADS), the medical society established in 1999 to provide scientific guidance to community doctors in the trenches of Lyme disease, published its own recommendations in 2004, and then again in 2014, to treat Lyme disease with a longer, four- to six-week antibiotics course of doxycycline, amoxicillin, or cefuroxime. The ILADS guidelines also recommend reassessing patients after their initial antibiotics therapy and, when necessary, extending it. ILADS based its recommendations on studies that identified the presence of the bacterium in animals post-antibiotics treatment, conferring legitimacy on the theory of persisting infection. In that spirit, ILADS members have experimented with combination antibiotics therapies and treatment sustained for as long as symptoms persist.
The stakes were high not only for the patients but also for the credibility and reputation of the professionals who dared make treatment recommendations or decisions without definitive scientific knowledge. For more than 30 years, the two sides have engaged in what medical doctors Raphael B. Stricker and Andrew Lautin coined the “Lyme Wars,” a contentious debate about the biological basis and existence of late-stage Lyme. The entrenched hostility between the two camps sowed both distrust in the science and confusion among patients who were desperate for answers. Leaders of these organizations played a role in fostering this distrust. As Brian Fallon, the director of the Lyme and Tick-borne Diseases Research Center at the Columbia University Medical Center, and Jennifer Sotsky, a resident at the Columbia University Irving Medical Center, explained in their 2017 book, Conquering Lyme Disease: Science Bridges the Great Divide, “[They have taken] entrenched positions, refusing to budge, focusing on absolutes rather than acknowledging the areas of uncertainty, selectively pulling journal articles from the published literature that favor their position and not presenting those articles that suggest otherwise.”
As this unproductive debate unfolded in the 2000s, the Centers for Disease Control and Prevention (CDC) adopted a narrow definition of Lyme disease for the exclusive purpose of surveillance. This step resulted in the CDC’s underestimating cases by a factor of 10, as it admitted in a 2021 article. Two decades of underreporting masked the growing epidemic and justified the absence of a dedicated tick-borne-disease curriculum in medical schools, all while barriers to care remained extremely high. The false sense of low prevalence also created the illusion of small markets and poor economic incentives to invest in solutions to Lyme disease. Subsequently, throughout the first two decades of the 21st century, university and industry investments in Lyme disease science remained minimal, delaying the development of diagnostics and therapeutics that accurately tested, detected, and treated all stages of the disease.
A Citizen Movement Emerges
When Lyme’s scientific progress hit an impasse, the community of patients felt increasingly abandoned by the medical establishment. “Back in the 2000s, most community groups were focused on helping patients get the care they deserved,” says Bay Area Lyme Foundation executive director Linda Giampa. Tight-knit communities of patient advocates—primarily in high-incidence states on the East Coast—served patients through support groups and prevention programs. Some of them also successfully petitioned their state legislators to establish public-education mandates and legal protection for doctors who ventured to treat late-stage Lyme cases. Yet patients continued to endure the physical pain and financial ruin resulting from inadequate and costly treatments not covered by health insurance. Their desperate cries for help sometimes added oil to the fire already raging between the two Lyme camps. As a result, they experienced stigmatization and ostracization that profoundly affected their access to care.
 Northern Arizona University assistant professor in biological sciences Nate Nieto performs a tick sweep in Marin County, California, for a 2017 Bay Area Lyme Foundation tick study. (Photo courtesy of Bay Area Lyme Foundation)
Northern Arizona University assistant professor in biological sciences Nate Nieto performs a tick sweep in Marin County, California, for a 2017 Bay Area Lyme Foundation tick study. (Photo courtesy of Bay Area Lyme Foundation)
The divide between patients and professionals was bridged in the mid-2010s, when a handful of highly respected scientists and doctors contracted Lyme disease. These patients included Neil Spector, an oncologist and cancer researcher; Steven Phillips, principal investigator for Kyronyx Biosciences and a renowned expert on zoonotic infections; Alan McDonald, a pathologist; and Richard Horowitz, a physician and a founding member and former president of ILADS. They made critical contributions to our understanding of Lyme pathology in their effort to reconcile their theoretical understanding with their lived experience. When their respective clinical and academic trainings anchored in the nonpersistence model failed to restore them to full wellness, they started to recognize the merits of both disease models. Opening to the possibility of pathogen persistence, they started painting a much more complex picture accounting for the diversity of the pathogens and for the multiple possible disease pathways.
Supported by these new allies from the medical and scientific communities, patient advocates redoubled their efforts with new hope and fresh thinking. They quickly realized that a coordinated nationwide effort leveraging federal infrastructures and resources to tackle the threat of tick-borne diseases strategically and methodically was missing. In particular, the pipeline of scientists dedicating their careers to tick-borne diseases around the country did not exist.
“We desperately needed to attract new scientists and fresh perspectives to the Lyme space in order to develop an accurate diagnostic,” Giampa says. “For that, we had to remove existing barriers to research.”
Since National Institutes of Health (NIH) funding for Lyme disease was scarce, the Bay Area Lyme Foundation and the Steven & Alexandra Cohen Foundation stepped in with private funding. They have fueled Lyme research with more than $100 million in grants in the past decade. In 2014, the Bay Area Lyme Foundation launched the Emerging Leader Award to direct some of this funding to new investigators by providing a financial incentive to start new research labs. Beyond addressing the funding gap, the foundations collaborated to build the Lyme Disease Biobank, a repository of blood, urine, and human-tissue samples that enables scientists to easily access samples they need for their experiments.
In 2015, the Bay Area Lyme Foundation created Lyme Innovation, a multistakeholder initiative to inventory knowledge gaps, develop priorities, and deploy financial incentives to attract innovators to solve the most consequential problems of Lyme disease. This initiative was led by Kristen Honey, a fellow at Stanford University’s Robinson Lab headed by immunologist and rheumatologist William H. Robinson, and Nevena Zubcevik, the cofounder of the Dean Center for Tick Borne Illness at the Spaulding Rehabilitation Hospital, an affiliate of Harvard Medical School.
Wanting to move beyond the Lyme Wars, the group was committed to revolutionizing the scope and nature of Lyme research by being open to considering all scientific hypotheses. “The field had been stuck in a false dichotomy,” Zubcevik says. “Both models offered unique pieces to a complex puzzle. The time was ripe for an intervention that would propel Lyme research beyond the persistence debate and bring much needed solutions to Lyme patients.”
Coming together to organize around shared goals, Lyme Innovation founding partners included reputable organizations, such as the Dean Center for Tick Borne Illness, the Bay Area Lyme Foundation, MIT Hacking Medicine, and the Veterans Affairs Center for Innovation to Implementation. They also embraced an eclectic group of independent volunteers of university affiliates and Lyme patients who participated thanks to the support of the American Association for the Advancement of Science and Technology Policy Fellowship program.
Between 2016 and 2017, Lyme Innovation conducted a hackathon-style innovation process in partnership with academic and nonprofit organizations on both coasts, designed to attract and empower problem solvers from academic disciplines and professions not yet tapped to solve Lyme issues.
“I came to [the field of] Lyme from the environmental sciences, and it was clear to me that people from outside the medical community—whether patients or industry leaders—had much outside-the-box thinking to contribute to the field,” Honey says. The hackathon events allowed participants to access Lyme insights from scientific experts and to learn from those affected by the disease. “We operated with an open ethos and a user-centric approach,” she explains of the process. “Those with lived experiences worked side by side, as equals, with physicians, scientists, and decision makers in both science and policy discussions.”
This community-driven process brought new researchers into the field and resulted in more than two dozen new Lyme-related research projects and initiatives, including G-Dot, a project that explored whether a therapeutic drug in clinical trials for cancer could block Borrelia from DNA recombination; LymeDot, a tracking system for patients to record their symptoms; and CrisisText Lyme, a platform that combated isolation, depression, and suicidal feelings among Lyme patients by providing a text-based crisis line.
 A woman provides a Lyme disease biobank donation at the San Diego collection site, in March 2020. (Photo courtesy of Bay Area Lyme Foundation)
A woman provides a Lyme disease biobank donation at the San Diego collection site, in March 2020. (Photo courtesy of Bay Area Lyme Foundation)
The movement served as an informal locus of coordination for the Lyme community until 2018. With the Bay Area Lyme Foundation as a convener and sponsor, patient advocates came together in roundtable exercises to develop an understanding of the steps needed to achieve their shared goal of advancing the detection and treatment of Lyme disease. Organizations contributed new programs to a grand strategy that pursued the movement’s priorities.
Today, LymeDisease.org leads national prevention campaigns to slow the pace of the tick-borne-disease epidemic by raising awareness about the risks associated with outdoor activities. It also runs MyLymeData, a patient-led research effort that documents patients’ experience of the disease. Other organizations at the front lines include the Lyme Disease Association, which operates a doctor-referral database and advocates for legislation that promotes scientific research; LymeLight Foundation, which provides financial support for current patients in the absence of health-insurance coverage; the Center for Lyme Action, which educates US congressional representatives about the urgency to increase resources for Lyme disease prevention and research; Invisible International, which builds evidence-based continuing medical education modules for physicians and clinicians; and the Bay Area Lyme Foundation and Steven & Alexandra Cohen Foundation, which fund scientific research to make Lyme easier to diagnose and cure.
Building Governmental Infrastructure
The relationship between the government and the community of Lyme patients was fraught because patients’ interests were at odds with HHS’ public-health agenda. Indeed, in 2015 the CDC published a National Action Plan for Combating Antibiotic-Resistant Bacteria, which sought to improve antibiotics stewardship in health-care settings yet conflicted with the community’s desire for longer antibiotics treatments. More important, patient advocates resented the outsize influence that the mainstream medical establishment—which they disagreed with—exercised in administrative offices. IDSA members, who belonged in the nonpersistence-hypothesis camp, heavily staffed the NIH’s scientific-grant review boards and thwarted research proposals that sought funding to explore alternative disease hypotheses. In 2012, Stephen Barthold, a professor of medical pathology at the UC Davis School of Veterinary Medicine and the director of the UC Davis Center for Comparative Medicine, testified before a House of Representatives committee about the difficulty of the NIH grant process when applying for funding for his work on the persistence of Borrelia in animal models. Representation of more diverse Lyme disease viewpoints in the peer-review process for government grants was sorely needed to unleash the full innovation potential of the research community.
In a decade-long advocacy effort beginning in the mid-2000s, several Lyme-disease nonprofits led by LymeDisease.org worked with the US Congress to establish a balanced advisory committee that could expertly guide the creation of a federal program to address the growing epidemic. The effort culminated in December 2016 with the passage of the 21st Century Cures Act, which mandated that HHS establish a federal advisory committee called Tick-Borne Disease Working Group to evaluate its existing research efforts related to tick-borne diseases. Wendy Adams, the Bay Area Lyme Foundation’s research grant director and a member of the Working Group from 2017 to 2020, says that every two years the group was required to provide Congress and the HHS Secretary with “an objective account of the issue, its prevalence, the state of the scientific knowledge” and to “make priority recommendations for resource allocation.”
The founding charter of the working group mandated diversity of thought among its governmental and nongovernmental members to more purposefully bridge the divide between stakeholders. “The Lyme debates were still raging, and our credibility hinged on the inclusion of government officials, doctors, scientists, and patients, with differing perspectives on the best approach to the issues,” Adams says. “It was the first time [that] patients had a meaningful seat at the table.” Responding to the call for diverse participants, several members of the Lyme Innovation movement joined the working group. While it took almost 50 years after the first Lyme cluster was identified, the Lyme community finally had a voice at the national level and a process to access greater resources.
The working group wrestled with fundamental questions that had torn the Lyme world apart for years. The most important and contentious question continued to be whether Borrelia can persist after antibiotics treatment—in other words, whether chronic Lyme exists. The group recognized that a significant percentage of patients, treated or untreated, have persistent symptoms.
“The absence of any definitive means to determine if the causative organisms are still present or absent in such patients made it impossible to understand these symptoms,” explains IDSA member and working-group participant Sam Donta. Therefore, in its 2020 report, the clinical subgroup that Donta chaired requested that the US Congress “provide appropriate funding to better understand the pathophysiology of the disease, to discover means for the direct detection of persisting organisms or their products, and to develop treatment regimens that can cure patients of their persistent disease.”
Congress subsequently increased HHS appropriations for tick-borne-disease research from $34 million in 2020 to $81 million in 2021, and then raised it to $114 million in 2022. Despite these much needed increases, Lyme disease research remains underfunded ($82 per patient) compared with other infectious diseases with lower prevalence in the United States, such as the West Nile virus ($6,870 per patient) and malaria ($150,500 per patient).
The working group set the stage for a new era in Lyme research beyond the persistence/nonpersistence divide. To usher in a new wave of research, HHS experimented with a new grant-allocation process designed specifically to open the breadth of scientific exploration while focusing resources on the greatest needs. In 2018, HHS launched the HHS Lyme Innovation Initiative, incorporating and expanding on the successful community-led Lyme Innovation model. Honey, who was HHS Lyme Innovation’s first administrator, served as a coordinator across governmental entities and continues to convene the various stakeholders of the Lyme community.
In October 2020, HHS Lyme Innovation launched a $25 million LymeX Innovation Accelerator in partnership with the Steven & Alexandra Cohen Foundation to scale the Lyme Innovation hackathon’s model. As the largest private donor for Lyme disease research in the United States, the foundation funded the infrastructure necessary to continue to refine innovation priorities in collaboration with Lyme stakeholders and to incentivize the development of new solutions in the areas of tick-borne-disease education and diagnostics.
To address the need for effective diagnostics, LymeX launched a competition in 2022—the LymeX Diagnostics Prize—with up to $10 million in cash prizes across multiple phases for the next generation of technologies to detect active Lyme infections in people. The LymeX Diagnostics Prize judging panel selected 10 winning teams in the first phase of the competition, which enabled them to proceed with subsequent steps in their projects, including prototyping, data validation, and clinical-protocol development. LymeX will continue to support the best teams among the initial winners in subsequent stages of the competition.
Bigger Than Lyme
The work of creating responsive health-care infrastructures continues beyond Lyme disease to all vector-borne diseases and to complex chronic diseases more broadly.
The working group successfully established itself as Congress’ legitimate source of expertise on tick-borne diseases. Therefore, when the 2019 Kay Hagan Tick Act mandated that HHS create a national strategy to address vector-borne diseases, the Office of the Assistant Secretary for Health (OASH) and the CDC requested the working group as their collaborator in a three-year effort that culminated in the publication of the first-ever document of its kind, in January 2024. The strategy outlines five goals: understanding the pathogenesis, refining diagnostic tools, developing treatments, strengthening prevention, and supporting public health. It incorporates community input along 19 strategic priorities that the federal government developed using the 2020 National Public Health Framework for the Prevention and Control of Vector-Borne Diseases in Humans. It offers a striking acknowledgment of vector-borne diseases as a threat to human health and a helpful road map to stimulate and orchestrate the national response to these very specific forms of acute and chronic diseases.
Under the influence of ongoing pathological, environmental, and psychological stressors, our immune response may click into overdrive and produce harmful inflammatory symptoms that are the hallmark of many chronic diseases. When our survival response becomes permanently turned on, our offspring are likely to inherit the inflammatory expression of our genes.
However, in the four years it took to produce the strategy, the COVID-19 pandemic drastically transformed our understanding of chronic diseases. Today, late-stage Lyme, myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), long COVID, and postural orthostatic tachycardia syndrome (POTS) affect 18 to 23 million Americans—a number that has substantially increased as a result of the pandemic and is still growing. Late-stage Lyme disease and ME/CFS sufferers were quick to recognize their symptoms in the list of complaints that long-COVID sufferers reported; symptoms among people affected by these conditions are similar in their multisystemic, fatiguing, nonspecific, chronic nature.
“How you got to Rome may not matter as much as the fact that you are in Rome,” says MIT scientist Michal Tal. “Whether you got there on a bacterial road via Lyme disease or a viral road via COVID, the important thing, once you’re in Rome, is how to get you back out.”
Understanding the shared underlying biological features of these conditions may provide crucial insights about how to solve them. Among these shared features, chronic diseases may be prompted by an infection, are the expression of an overwhelmed immune system, and affect women more than men. An increasing number of scientific studies demonstrate a connection between chronic diseases and infections. The pathogenesis of Lyme disease, for example, has been established as a bacterial infection, and that of long COVID as a viral infection. Similarly, symptoms of fibromyalgia may start after a variety of infections, and scientists believe that the Epstein-Barr virus causes some cases of ME/CFS, lupus, multiple sclerosis, and many other autoimmune diseases.
The recognition that infections are a causal agent of chronic diseases has resulted in the development of new models of disease that depart from the predominant 19th-century germ theory, which reduces diseases to simple interactions between pathogens and the human host. Rather, these models emphasize the variability of human response to infections and the interaction between the pathogen, the immune system, and the environment to explain why some patients swiftly overcome diseases while others may develop autoimmune conditions, chronic fatigue, and pain over time. The new models relegate germs to simple triggers and account for the influence of genetic, social, and environmental factors in cultivating the biological terrain that informs the human immune response. Far from diagnostic certainty and miracle cures, these models encourage prevention and the mitigation of an array of immune distress risks at a time when signs of disease may not yet be visible.
 Chronic disease patients must often adopt regimens of numerous supplements.
Chronic disease patients must often adopt regimens of numerous supplements.
Furthermore, whether infections overwhelm or disregulate the immune system, complex chronic conditions almost always involve a failure of the immune system to manage the ecosystem of microbes teeming inside the human body. Some of the most dangerous infectious agents in human health have developed immune-evading tactics: Some, for example, enter a dormant state that lower their vulnerability to treatment; others develop colonies behind an impenetrable biofilm that enables them to subvert our defense mechanisms.
The impact of these sophisticated pathogens is compounded by a human immune system evolutionarily unprepared for the speed of societal change since the industrial revolution. For example, sudden changes in the human microbiome compromise its ability to train and develop the major components of both the innate and the adaptive immune systems. The higher prevalence of chronic diseases and concomitant relative paucity of microbiome diversity in the Western world today, compared with contemporary hunter-gatherer communities, who opted out of modernization, reveals the consequences of the Western lifestyle: The standard American high-fat diet, reliance on antibiotics, and overuse of antiseptics have disrupted the Western body’s gut flora, resulting in low diversity and therefore in poorly trained immune defenses. Food processing has increased intestinal permeability and compromised the blood-brain barrier, forcing the immune system to mount a response to the partially digested foods that find their way into the bloodstream. This “leaky gut/brain” phenomenon has been linked to food sensitivities, allergies, and autoimmune reactions.
In addition to a compromised microbiome, today’s immune system must contend with toxic exposures from birth. A 2005 Environmental Working Group study conducted by two major laboratories (AXYS Analytical Services Ltd. and Flett Research Ltd.) revealed the presence of 287 industrial chemicals in the fetal cord blood of ten newborns. Chronic-disease patients are routinely tested, diagnosed with, and treated for heavy-metal and pesticides toxicity—all new, man-made, toxic substances that did not exist 100 years ago and that the modern immune system now needs to identify as foreign and eliminate.
Under the influence of ongoing pathological, environmental, and psychological stressors, our immune response may click into overdrive and produce harmful inflammatory symptoms that are the hallmark of many chronic diseases. “Our bodies are not just trained to survive but to survive at any cost,” writes holistic-medicine practitioner Isaac Eliaz in his 2021 book, The Survival Paradox. “In an attempt to survive a stressor, our survival response may cause more damage than the stressor itself.” When our environment and the rapid pace of our lives constantly prompt a survival response, the epigenetic layer of molecules that governs the readability of our DNA adapts to the need for ongoing activation of the sympathetic nervous system. In other words, while it takes several generations for DNA to evolve, it takes only a few years and the right lifestyle and environmental conditions to transform our gene expression. When our survival response becomes permanently turned on, our offspring are likely to inherit not only our deficiencies and toxicities but also the inflammatory expression of our genes. The reality is that US society is already in the process of an epigenetic shift toward the sympathetic dominance of the immune function: Chronic conditions affect 31 percent of children under the age of 18 and approximately one-half of young adults ages 18 to 34 in the United States.
Complex chronic conditions also show a stronger prevalence among women than men, who comprise 58 percent of patients with Lyme, 85 percent with POTS, 80 percent with ME/CFS, and 80 percent across all autoimmune conditions. The patient, medical, and scientific communities have established several hypotheses for this gender disparity: Biological factors include differentiated immune responses to infections, the role of hormones and estrogen in symptomatology, and the role of pheromones in attracting pathogens. Social and cultural factors include the lack of therapies responsive to women’s bodies in a world where drugs are tested mostly on male humans and animals, and the medical profession’s tendency to systemically dismiss women’s pain, which can prevent a prompt and accurate diagnosis.
The New Fight
Infection-associated complex chronic diseases have been rising to new levels of collective consciousness since the beginning of the COVID-19 pandemic. Scientists, doctors, and patients are eager to push the frontiers of knowledge and to transform our medical and governmental infrastructures to address this emerging health crisis.
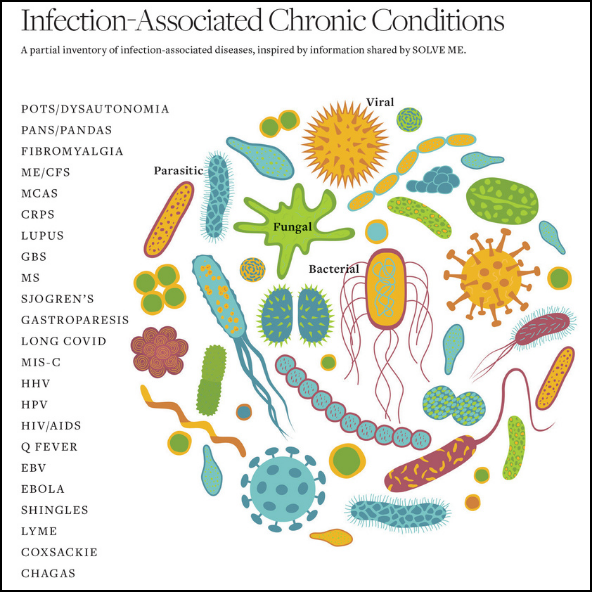
The scientific community is tuned in to the shared reality of long-haul diseases and is actively trying to understand the biology that underlies these shared symptoms. Last June, a National Academies workshop cohosted by LymeDisease.org’s CEO, Lorraine Johnson, convened high-level health officials, prominent researchers, and patient advocates from a variety of conditions to study common, overlapping clinical and biological factors among infection-associated chronic illnesses. They examined the results of patient-led research on long COVID and Lyme disease, as well as the latest academic research on immune dysfunction, autonomic dysfunction, brain inflammation, and the role of the microbiome, among many other sessions. It was a rare opportunity for scientists and other concerned professionals to compare symptoms and leading hypotheses to explain these diseases.
The size and complexity of the research projects required to make sense of complex chronic conditions are significantly greater than those for single-organ pathologies. “Research across specialties has become critical,” Giampa explains. “We need studies that allow the inclusion of a broad set of case definitions across multiple diseases while shortening the time from lab to bedside.” Research exploring the interconnectedness and the causality between what were previously thought to be separate conditions is particularly difficult. One example is the current effort to understand the connection between chronic infections and neurodegenerative diseases. In 2023, molecular biologist Richard Lathe, Intracell Research Group founder Nikki Schultek, and Philadelphia College of Osteopathic Medicine professor of neuroscience and neuropathology Brian Balin, among other experts, called for international scientific collaboration to investigate how microbial infections such as Lyme neuroborreliosis—the infection of the brain by Borrelia—may cause neurodegenerative diseases. The collaborative proposed the establishment of a consensus protocol to explore the brain pathobiome—the community of microbes inside the brain—in patients with dementia and/or Alzheimer’s disease.
While scientists look for answers, doctors remain on the front lines of the public-health response to human suffering. They too have come to appreciate the similarities between complex chronic conditions and are building on lessons learned from Lyme disease to develop supportive protocols for complex chronic conditions more broadly. “In scarcely one and a half generations of doctors, Lyme disease has gone from being a newly recognized bacterial infection to becoming the standard of comparison for approaching chronic complex illnesses,” says ILADS president-elect Steven Harris. “The entire gamut of medicine and medical science is employed to address this disease model.”
Before the pandemic accelerated the prevalence of complex chronic conditions, ILADS had expanded its mission to support doctors in their work treating them. The ILADS board is even considering changing the name of the organization to reflect the breadth of relevant medical issues and to honor ILADS’ insights about the nature of persistent symptoms. The organization has extended its evidence-based fellowship credentialing from community doctors in the trenches of Lyme disease to doctors working with the chronically ill more broadly, and it will continue to contribute to the advancement of biomedicine by studying, codifying, and disseminating emerging treatment approaches.
The existing medical infrastructure is inadequate to coordinate the multidisciplinary care that chronic diseases demand. “To address the challenge of complex chronic diseases, we must stop looking at them as separate conditions and build a new generation of medical institutions that facilitate collaboration between medical disciplines and break down barriers between scientific research and patient care,” says medical doctor Laura Pace. She founded the Metrodora Institute, a clinic dedicated to neuroimmune axis disorders, as a blueprint to realize this vision.
It’s time for all relevant disease communities of patients, doctors, and scientists to start working together and collaboratively with our government representatives on a new, even more ambitious national strategy that can provide academics and industry leaders with a framework to address infection-associated chronic diseases.
The increase in the number of long-COVID patients provides a strong incentive for medical institutions to create new structures capable of delivering medical care across multiple medical specialties. “The world’s attention is on COVID and its aftereffects in ways that it never was on Lyme disease or ME/CFS,” chronic-disease patient Meghan O’Rourke observes in her 2022 book, The Invisible Kingdom: Reimagining Chronic Illness. “Major medical institutions swiftly acted on the need for an organized approach to infection-associated chronic illnesses. We have an opportunity to build the medical infrastructure we need to address all chronic illnesses, not just long COVID.”
Members of the patient community are also eager to support one another across individual disease communities and to contribute their shared experience to facilitate progress on medical solutions and patient accompaniment. Because symptoms can be quickly miscategorized as psychosomatic, long-haul-disease patients are often dismissed as anxious or depressed. For fear of being misunderstood, the patient community started organizing across multiple conditions. In 2023, a new Infection-Associated Chronic Conditions Patient Advocacy Coalition (IACC-PAC) brought together patient-advocacy organizations to champion the collective needs of the infection-associated, chronic-condition community. Together, they are fighting for the inclusion of patients’ voices in scientific research and to inform the public-health agenda.
High on their list of recommendations is the creation of appropriate government infrastructures. “We need an NIH office dedicated to the growing number of complex chronic conditions that can accelerate multidisciplinary approaches and interagency collaboration while creating new opportunities for patient engagement and stakeholder input,” says Emily Taylor, the vice president of advocacy and engagement at SOLVE M.E., a nonprofit working to help people living with ME/CFS, and an IACC-PAC member. A series of recent IACC-PAC workshops generated additional priorities, including health-insurance coverage, disability benefits, simplified coordination of care, greater cultural acceptance in workplace and educational settings, and physician training.
Moving Forward Together
The Lyme innovation movement and its collaboration with the federal government provided the necessary spark and resources to develop a national strategy for vector-borne diseases. However, Lyme disease was only one manifestation of the much larger medical challenge presented by infection-associated complex chronic diseases. The holistic understanding of these conditions is gaining momentum in the scientific, medical, and patient communities. Phillips predicts that their recognition as a unified medical phenomenon “augurs a fundamental reset of every aspect of societal response.”
It’s time for all relevant disease communities of patients, doctors, and scientists to start working together and collaboratively with our government representatives on a new, even more ambitious national strategy that can provide academics and industry leaders with a helpful framework to address infection-associated chronic diseases across the many complex and elusive conditions that affect people today.
As we go about this work, we can draw lessons from the battles we fought in the past, the mistakes we made, and the factors that were critical to our successes. Let’s remember how important it is to stay open to all scientific hypotheses to unleash the full innovation potential of our nation. Let’s stay committed to diversity of thought in administrative bodies so our government institutions can truly fulfill their function on behalf of the people. Let’s bring together those closest to the problem and those controlling decisions and resources into a cocreation process to make sure it doesn’t take another 50 years to address infection-associated complex chronic illnesses.
Read more stories by Bernadette Clavier.


